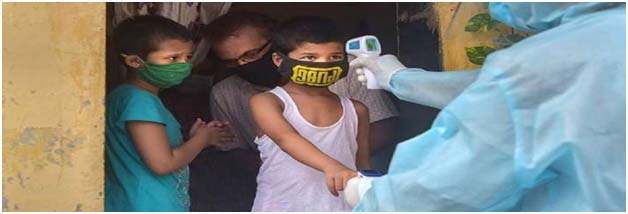
New Delhi: Wednesday din India laga central drug authority, expert panel para Serum Institute of India (SII) logot phase 2/3 trial COVID-19 vaccine Covovax toh bacha 2 saal umer para 17 umer logot manishe, koikena khobor para janaishe. Monday din te Serum Institute of India para Drugs Controller General of India (DCGI) logot 920 bacha ke trial loboli hotishe aro etu 10 jaga te trail toh koriboli ase koishe. COVID-19 Central Drugs Standard Control Organisation (CDSCO)Subject Expert Committee (SEC) laga Subject Expert Committee (SEC) para application te vaccine toh desh te aro approved hoikena nai”, koikena khobor para janaishe. “etu oper te aro koishe ke, Pune-based company para safety & immunogenicity data (of Covovax) toh ongoing clinical trial loi thaka toh submit koriboli janaishe aro bacha khan ke clinical trial loboli koishe,” khobor para koishe. Etu recommendations toh DCGI para approved kora ase. August 2020 saal te, US-based vaccine maker Novavax Inc.para licence agreement toh Serum Institute of India ke development & commercialisation NVX-CoV2373, COVID-19 vaccine candidate toh low and middle-income countries aro India logot bonaikena ase koishe. India te clinical trials of Covovax toh March moina te soruh hoishe aro Serum Institute of India para jawan manuh nemite September moina te kuliboli ase koishe. January moina te, Serum Institute of India para Covishield vaccine toh desh te olaishe aro University of Oxford arp AstraZeneca vaccine bona company logot melikena ase koishe.
Govt para bacha 2-17 umer ke Covovax trial manishe
July 1, 2021
-
Share This!








Add Comment